Letã¢â‚¬â„¢s Make Microglia Great Again in Neurodegenerative Disorders
- Review
- Open Access
- Published:
Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes
Translational Neurodegeneration book 9, Article number:42 (2020) Cite this article
Abstract
Neuroinflammation is associated with neurodegenerative diseases, such as Alzheimer'south illness, Parkinson's disease, and amyotrophic lateral sclerosis. Microglia and astrocytes are key regulators of inflammatory responses in the central nervous system. The activation of microglia and astrocytes is heterogeneous and traditionally categorized as neurotoxic (M1-phenotype microglia and A1-phenotype astrocytes) or neuroprotective (M2-phenotype microglia and A2-phenotype astrocytes). All the same, this dichotomized classification may not reflect the various phenotypes of microglia and astrocytes. The relationship between these activated glial cells is as well very complicated, and the phenotypic distribution tin modify, based on the progression of neurodegenerative diseases. A better understanding of the roles of microglia and astrocytes in neurodegenerative diseases is essential for developing constructive therapies. In this review, nosotros discuss the roles of inflammatory response in neurodegenerative diseases, focusing on the contributions of microglia and astrocytes and their relationship. In addition, we discuss biomarkers to measure out neuroinflammation and studies on therapeutic drugs that can modulate neuroinflammation.
Background
With the increase in life expectancy, the global socioeconomic impact of neurodegenerative diseases, including Alzheimer'southward disease (Advertisement), Parkinson's illness (PD), and amyotrophic lateral sclerosis (ALS), is increasing considerably [1]. However, the pathological mechanisms underlying neurodegenerative diseases are non fully understood. Several factors including genetic, environmental, and endogenous factors are involved. Abnormal protein dynamics, oxidative stress with reactive oxygen species, mitochondrial dysfunction, Dna damage, dysfunction of neurotrophins, and neuroinflammatory processes are considered to be mutual pathophysiological mechanisms [ii]. Neuroinflammation is a defense machinery that initially protects the brain by removing or inhibiting various pathogens [3]. This inflammatory response tin accept benign effects by promoting tissue repair and removing cellular droppings. Sustained inflammatory responses, however, are detrimental, and they inhibit regeneration [4, 5]. Inflammatory stimulation can persist due to endogenous (eastward.g., genetic mutation and protein aggregation) or environmental (due east.k., infection, trauma, and drugs) factors [half-dozen, vii]. The persistent inflammatory responses involve microglia and astrocytes and can lead to neurodegenerative diseases [4].
Two categories of cells populate the cardinal nervous organisation: neurons and glial cells [8]. Glial cells do not produce electrical impulses, and they were considered as supporting cells for neurons. It has been revealed that glial cells are superior to neurons in cellular variety and function [9]. Glial cells, including astrocytes, oligodendrocytes, and microglia, can regulate neuronal activity [viii, ten]. Microglia and astrocytes serve diverse functions including innate immune responses in the encephalon. Traditionally, both can be classified into two opposing phenotypes: neurotoxic and neuroprotective. Microglia are divided into the M1 (classical activation) and M2 (alternative activation) phenotypes based on their activation condition [6, eleven]. Similar to the microglia, astrocytes can produce pro-inflammatory or immunoregulatory mediators co-ordinate to the phenotype of the polarization status [7]. Notwithstanding, microglia and astrocytes are considered to take multiple reactive phenotypes related to the type and stage of neurodegenerative diseases and the regional location [12,13,14]. Furthermore, the changes in phenotypes of microglia and astrocytes, their loss of neuroprotective functions, and their gain of neurotoxic functions are complicated and may differ with the phase and severity of neurodegenerative diseases. Therefore, the elementary dichotomized classification cannot reverberate the various phenotypes of microglia and astrocytes [12]. For these reasons, the utilize of the M1/M2 and A1/A2 nomenclature was limited in this manuscript, appearing only directly from the references where they were used. However, they should be considered equally being on a spectrum, rather than being two distinct populations. This complexity could be the reason why trials of anti-inflammatory drugs accept, to date, failed to show pregnant therapeutic effects. Here, we review the roles of inflammatory responses in neurodegenerative diseases, such as Advertising, PD, and ALS, focusing on the roles of microglia and astrocytes and their relationships. Recommendations for the success of clinical trials are too made. In addition, biomarkers to measure neuroinflammation and studies on drugs that can attune neuroinflammation are also discussed.
Microglia
Microglia are ubiquitously distributed in the encephalon and are the principal innate immune cells and the first responders to pathological insults [15, 16]. The proportion of microglia ranges five–12% of the total prison cell population in the mouse brain depending on the location, and they accept diverse morphologies: compact circular, longitudinally branched, and radially branched [17]. They are involved in homeostasis and host defense mechanisms past participating in 3 essential functions [18]. The first function is detecting changes in their environment using their sensomes, which are encoded past various genes [xix]. The 2d is the physiological housekeeping function, which includes migrating to injured sites, remodeling synapses, and maintaining myelin homeostasis [xviii, 20]. The 3rd is protecting against injurious stimuli, including pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs). Cellular receptors such as price-like receptors (TLRs), nuclear oligomerization domain-like receptors, and viral receptors are expressed on microglia, and tin can recognize PAMPs and DAMPs [half-dozen, 7]. In response to such stimuli, microglia produce proinflammatory cytokines, such as tumor necrosis gene (TNF)-α, interleukin (IL)-1β, IL-16 and chemokines, including the C-C motif chemokine ligand 2 (CCL2) and IL-18, to recruit additional cells and remove pathological agents [6, 18]. Even so, although neuroinflammation is a neuroprotective mechanism, sustained neuroinflammation tin induce neurotoxicity and is related to neurodegeneration [eighteen]. In addition, microglia priming with aging and chronic stress shows a dystrophic morphology and an exaggerated inflammatory response [21].
Microglial activation can exist assessed past imaging and fluid biomarkers. 11C-(R)PK11195 positron emission tomography (PET) can be used to quantify microglial activation via the binding chapters of 11C-(R)PK11195 to the translocator protein that is overexpressed in activated microglia [22, 23]. The soluble triggering receptor expressed on myeloid cells two (sTREM2), which is a cleavage product of TREM2 expressed on the cell surface of microglia [24, 25], is a fluid biomarker of microglial activation. Recent studies take shown that the cerebrospinal fluid (CSF) level of sTREM2 is correlated with plasma sTREM2 level, suggesting that the CSF sTREM2 is a potential biomarker for microglial activation [25, 26].
Microglia in the central nervous system (CNS) can exist pro-inflammatory or neuroprotective, depending on their activation status. Pro-inflammatory cytokines are droppings from pathogens or damaged cells, and they activate the resting microglia to express pro-inflammatory factors such as IL-1β, TNF-α, IL-6, nitric oxide (NO), and proteases, which have detrimental effects in neurodegenerative diseases (Fig. 1, Fig. 2) [6, 13]. In contrast, IL-four, IL-ten, IL-13, and transforming growth factor-β (TGF-β) activate neuroprotective microglia, which leads to the release of various factors including FIZZ1, Chitinase-three-Similar-three (Chi3l3), Arginase 1, Ym1, CD206, insulin-similar growth cistron 1 (IGF-1), and Frizzled form receptor ane (Fzd1) (Fig. 2) [vi, xiii, 27, 28]. These factors from microglia may exist associated with neuroprotection and tissue healing (Fig. 3). For instance, IL-four is known to suppress the release of pro-inflammatory cytokines, i.e., IL-half-dozen, TNF-α, and NO [29, xxx].
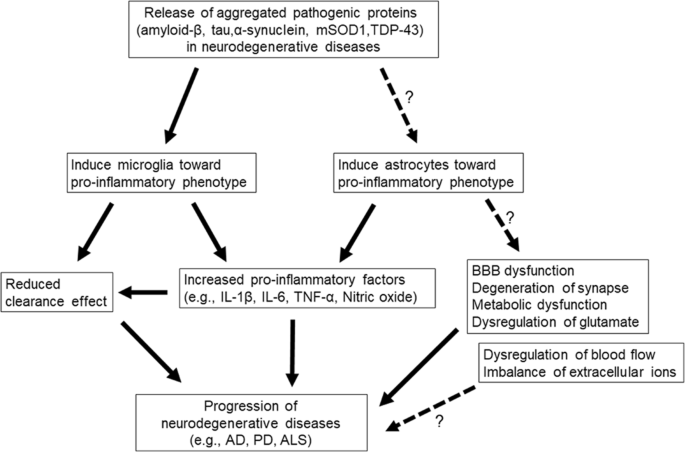
Potential relationships between neurodegenerative diseases and glial cells. The release of aggregated pathogenic proteins such as amyloid-β, tau, α-synuclein, mSOD1, and TDP-43, into the extracellular space drives the changes of microglia and astrocytes into their pro-inflammatory phenotypes. The predominance of the pro-inflammatory phenotype of microglia results in the increment of pro-inflammatory factors and a decrease of the phagocytic outcome. The pro-inflammatory-phenotype astrocytes release pro-inflammatory factors, which can dysregulate the synaptic function, the blood-brain barrier, metabolic role, glutamate, extracellular ions, and blood period. Ultimately, this tin can atomic number 82 to neurodegenerative disease progression. A dotted line with a question marker represents a possible relationship, with a lack of testify for a direct clan
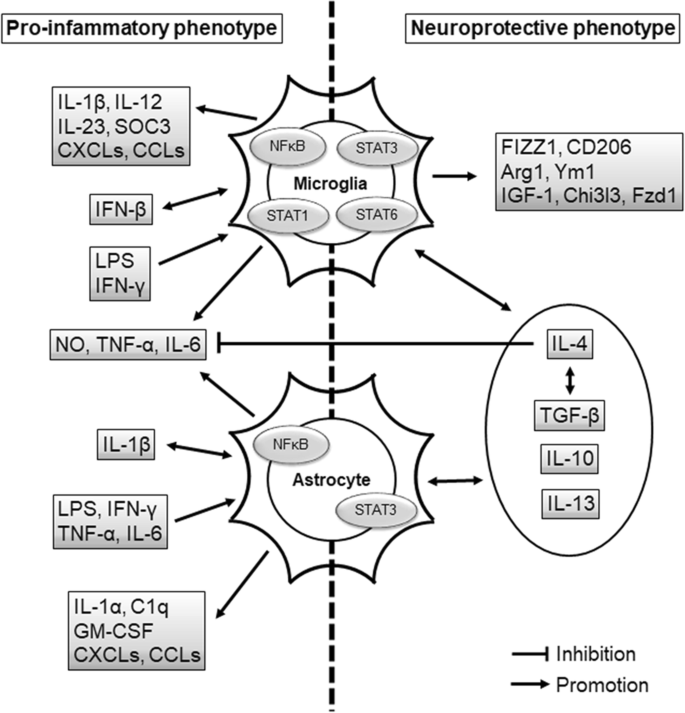
Proposed signals associated with microglia and astrocytes. The pro-inflammatory microglia are activated past IFNs and LPS via the activation of NFκB and STAT1, and then release IL-1β, IL-12, IL-23, SOC3, CXCLs, CCLs, NO, TNF-α, and IL-6. The neuroprotective microglia are promoted by IL-four, IL-xiii, IL-10, and TGF-β via the activation of STAT3 and STAT6. The M2 microglia enhance the neurotrophic factor (IGF-1), FIZZ1, CD206, Arg1, Ym1, Chi3l3, Fzd1, IL-13, IL-10, IL-4, and TGF-β. The activation of NFκB induces pro-inflammatory astrocytes. The pro-inflammatory astrocytes are afflicted past IL-1β, IFN-γ, LPS, TNF-α, and IL-6, and they produce IL-1α, C1q, GM-CSF, CXCLs, CCLS, TNF-α, IL-6, and NO. The activation of STAT3 induces neuroprotective astrocytes. The neuroprotective astrocytes interact with anti-inflammatory cytokines such as IL-thirteen, IL-x, TGF-β, and IL-4; IL-4 and TGF-β coordinate to promote protective effects, and IL-4 suppresses TNF-α, IL-vi, and NO. CCL: C-C-motif chemokine ligand; CXCL: C-10-C motif chemokine ligand; GM-CSF: granulocyte-macrophage colony-stimulating gene; IFN-γ: interferon γ; IL: interleukin; LPS: lipopolysaccharide; NFκB: nuclear factor κB; NO: nitric oxide; STAT: signal transducers and activators of transcription; TNFα: tumor necrosis factor α
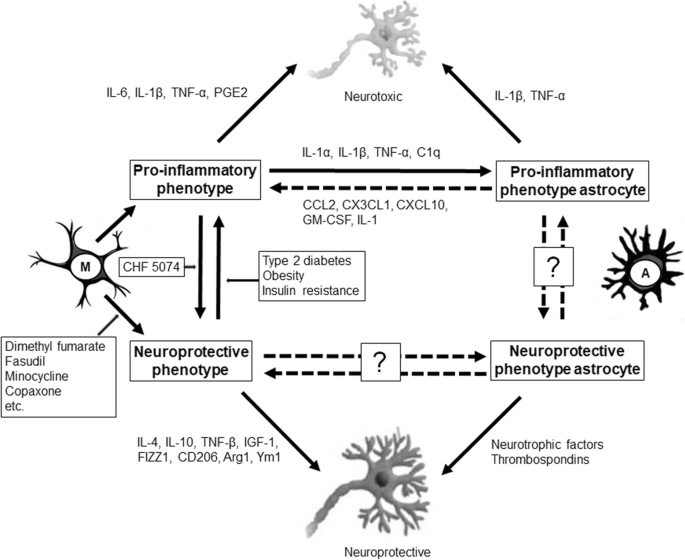
Schematic of microglial activation, astrocyte activation, and their relationship. The pro-inflammatory phenotypes are neurotoxic, while the neuroprotective phenotypes are neuroprotective. CHF 5074 polarizes microglia from the pro-inflammatory to the neuroprotective phenotype. Microglia tin can switch from the neuroprotective to the pro-inflammatory phenotype in the context of blazon 2 diabetes, obesity, and insulin resistance. Some candidates (dimethyl fumarate, fasudil, minocycline, and copaxone) tin potentiate the neuroprotective polarity of microglia. The pro-inflammatory microglia clandestine IL-1α, IL-1β, TNF-α, and C1q, which can modify astrocytes into the pro-inflammatory phenotype. The pro-inflammatory astrocytes underground CCL2, CX3CL1, CXCL10, GM-CSF, and IL-one, which in turn activate the pro-inflammatory microglia. The phenotype transition of astrocytes remains to be clarified. The dotted lines with question marks correspond a possible relationship, with a lack of evidence for straight association. M: resting microglia; A: astrocytes. Other abbreviations every bit in Fig. 2
Switching between these two phenotypes may touch remyelination, which is associated with aging [7, 31]. Obesity, insulin resistance, and type 2 diabetes are known to bear on the transition of microglia from the neuroprotective phenotype to the neurotoxic phenotype (Fig. 3) [28, 32, 33]. Several candidates and factors take been identified to potentiate the neuroprotective polarity: fasudil (Rho kinase inhibitor), Jumonji domain-containing 3 (Jmjd3, H3K27me3 demethylase), minocycline, Copaxone (glatiramer acetate), dimethyl fumarate (Tecfidera), cromolyn, CHF 5074, fingolimod, masitinib, glycogen synthase kinase-3 inhibitor, histone deacetylase inhibitor, peroxisome proliferator-activated receptor, adenosine monophosphate-activated protein kinase, and Janus kinase/bespeak transducers and activators of transcription (JAK/STAT) inhibitors (Fig. 3) [34,35,36,37,38,39]. The proportion of each phenotype tin can differ depending on the stage of neurodegenerative diseases [28]. Treatment targeting the phenotype residue may take different effects, depending on the time window [28]. Therefore, balancing and switching between the phenotypes of microglia at specific times and in specific patients may be important for modulating the progression of neurodegenerative diseases. The drugs that attune microglial activation are more likely to exhibit protective furnishings in a clinical trial that 1) utilise participants with more pro-inflammatory than neuroprotective microglial phenotypes, 2) enroll participants who are probable to bear witness progression within a few years, as it is difficult to follow patients for longer duration of years in clinical trials, and 3) have confirmed the pathology of the disease, such every bit amyloidopathy or tauopathy; without a pathological insult, the glial cells may non change. Further studies are warranted to investigate the appropriate time window and patients to demonstrate the clinical benefits of treatments targeting the microglial state.
Astrocytes
Astrocytes are the most common glial cells in the brain [40]. Although they were initially considered to but take passive functions, contempo studies have discovered that astrocytes play active and essential roles in brain homeostasis [41]. They regulate blood flow, maintain the blood-brain bulwark (BBB), provide energy metabolites to neurons, attune synaptic activity, control neurotrophin secretion, remove expressionless cells, too as regulating the extracellular balance of ions, fluid and transmitters, and scar formation [40,41,42]. Currently, glial fibrillary acidic poly peptide (GFAP), S100B, YKL040, and D-serine are assessed as CSF biomarkers and GFAP and S100B as blood biomarkers [43]. For imaging biomarkers, magnetic resonance spectroscopy, 11C-deuterium-L-deprenyl (11C-DED) PET, and 11C-BU PET are used to appraise the astrocyte reactivity [43]. Changes in molecular expression and morphology of astrocytes measured past GFAP can indicate the severity of reactive astrogliosis, which is a hallmark of CNS pathology [42]. Defects of astrocytes during the early on phase of injury including spinal cord injury (SCI) and experimental autoimmune encephalomyelitis (EAE), are consistently correlated with exacerbated clinical outcomes, neuroinflammation, BBB alteration, and neuronal decease [40], while on the other side, a written report in a chronic experimental EAE mouse model has shown that during chronic CNS inflammation, astrocytes produce lactosylceramide (LacCer), which promotes inflammation and neurodegeneration [44]. These results bespeak that the upshot of astrogliosis can be benign or detrimental, depending on the time, specific illness, and different stimuli from the microenvironment, such as microglia.
Astrocytes may accept multiple simultaneous reactive profiles, but with a continuous spectrum. Therefore, the heterogeneity of reactive astrocytes should be investigated further [xiii]. Similar to microglia, astrocytes accept pro-inflammatory and immunoregulatory (neuroprotective) subpopulations. The pro-inflammatory reactive astrocytes upregulate several genes (e.g., complement cascade genes) and induce pro-inflammatory factors (e.one thousand., IL-1β, TNF-α, and NO), which are known to have harmful functions (Fig. 1) [6, thirteen]. In comparison, the neuroprotective reactive astrocytes upregulate many neurotrophic factors and thrombospondins (Fig. iii) [13]. The anti-inflammatory cytokines, such as IL-4, IL-xiii, and IL-10, may induce neuroprotective activation of astrocytes, and these alternatively activated astrocytes may release IL-4, IL-10, and TGF-β (Fig. two) [41].
Inflammatory mediators secreted by pro-inflammatory microglia, such as IL-1α, IL-1β, TNF-α, and C1q, may actuate pro-inflammatory astrocytes and induce a secondary inflammatory response (Fig. 3) [45, 46]. Detrimental astrocytic signaling pathways can exist induced by several other cytokines, sphingolipids (sphingosine 1-phosphate and LacCer), and neurotrophins [40]. Astrocytes upregulate the transmembrane receptors for IL-17 and tropomyosin receptor kinase B (TrkB) during neuroinflammation. The binding of IL-17 to its receptors may upshot in the recruitment of nuclear factor κB (NFκB) activator one (Act1) and the production of pro-inflammatory cytokines [47]. Provisional mice lacking TrkB can be protected from EAE-induced neurodegeneration, while stimulation of TrkB past the agonist brain-derived neurotrophic factor (BDNF) has detrimental effects on neurons [48].
In contrast, astrocytes that respond to certain pathways are protective, since inhibition of the mediators of these protective pathways worsens neuroinflammation and neuronal cell death. The first protective pathway is mediated past glycoprotein gp130; it is related to the SHP2/Ras/ERK activation, and limits neuroinflammation [49]. A lack of gp130, a signal transducer for the IL-6 cytokine family, worsens the CNS injury of Toxoplasma encephalitis and EAE in mice [49, 50]. The 2nd protective pathway is mediated by TGFβ, which has important immunosuppressive properties. The astrocytic TGF-β signaling may mediate the inhibition of NFκB signaling and reduce neuroinflammation later on a stroke or Toxoplasma infection [51, 52]. The third protective pathway is mediated by interferon (IFN)-γ signaling. Although IFN-γ is a pro-inflammatory cytokine, the inhibition of its signaling in astrocytes worsens the mortality and leukocyte infiltration during the late phase of EAE in mice [53]. Finally, the estrogen receptor (ER) α signaling pathway in astrocytes has demonstrated anti-inflammatory and neuroprotective effects in diverse neurological affliction models [54].
The transcription factor indicate transducer and activator of transcription iii (STAT3) is expressed in astrocytes and phosphorylated subsequently injury. Ablation of STAT3 in astrocytes aggravates the infiltration of inflammatory cells, neuronal loss, and demyelination later on SCI in mice [55]. BDNF secreted past activated astrocytes can heighten STAT3 activation [56]. In another in vivo and in vitro written report involving an SCI mouse model, the knock-out of STAT3 adulterate astrogliosis and disrupted scar formation, which were associated with the worsening of inflammation and increased lesion volume [57]. Therefore, STAT3 seems to be critically involved in reactive astrogliosis, and is associated with a neuroprotective effect.
The JAK-STAT3 pathway may mediate the neuroprotective functions of reactive astrocytes. However, the molecular basis for the consecration of the neuroprotective reactive astrocytes is unclear [58]. In addition, there could be more than states of polarization than but proinflammatory or neuroprotective [13]. Therefore, the heterogeneity and molecular footing of reactive astrocytes should be investigated further.
Advertizing
AD is the most mutual class of dementia and is pathologically characterized by extracellular aggregating of amyloid-beta (Aβ)-containing plaques and evolution of intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein [16, 59]. In addition, neuroinflammation contributes to the pathogenesis of Advertising [16], as inflammatory responses have been repeatedly demonstrated in Advertizement. For instance, researchers have plant higher TNF-α (pro-inflammatory cytokine) and lower TNF-β (anti-inflammatory cytokine) levels in the CSF of balmy cognitive impairment patients who progressed to AD, compared with the controls who did not progress to AD [60]. Some cytokines including IL-1β, IL-six, and TNF-α have slowly increased levels from the early stage of the disease, while the levels of other cytokines including IL-18, MCP-1, and IP-ten tin can peak at a sure stage of the disease [61]. Although current publications take inconsistent results, it has been clear that neuroinflammation occurs early on in AD, and may trigger the progression of this affliction.
Morphological changes of microglia and astrocytes surrounding the senile plaques are also indicative of the neuroinflammatory response [half-dozen]. Both microglia and astrocytes collaborate with Aβ. Dysfunctions of microglia and astrocytic metabolism can effect in the accumulation of Aβ [18, 62]. Aβ in turn activates microglia and astrocytes through TLRs to release neuroinflammatory mediators that promote neurodegeneration [four, vi].
Microglia tin be neuroprotective past degrading and removing Aβ and tau [63, 64]. Even so, the persistent interaction betwixt Aβ and Aβ-induced pro-inflammatory cytokines overwhelms the clearance ability of microglia [18]. Increases in the size and number of Aβ plaques during the late-onset form of Advert may reflect the decreased clearance ability of microglia [65]. Microglia that surround the Aβ plaques are generally of the neuroprotective phenotype, labeled as Ym1, at the beginning of Aβ pathology; but they later switch to the neurotoxic (pro-inflammatory) phenotype during the avant-garde stage of the illness [28, 66]. The pro-inflammatory cytokines decrease the phagocytic activity of microglia, and they are likewise likely to transform microglia into the pro-inflammatory phenotypes. In improver, the pro-inflammatory microglia increment the phosphorylation of tau and exacerbate tau pathology [67]. This indicates that microglia in AD are involved in the Aβ and tau pathologies. In a recent study, microglial activation, every bit measured by elevenC-(R)PK11195, decreased longitudinally in patients with mild cerebral impairment and increased longitudinally in AD patients [68], suggesting that two peaks of microglial activation may exist nowadays in AD. Although the elevenC-(R)PK11195 cannot differentiate between the phenotypes of microglial activation, the first and terminal peaks may be neuroprotective and pro-inflammatory, respectively, equally microglia are known to change from the neuroprotective to the pro-inflammatory activation phenotypes during aging [68, 69]. In addition, the microglial activation is significantly correlated with amyloid degradation (measured by elevenC-PIB PET) [68], but not significantly correlated with tau accumulation (measured by eighteenF-AV-1451 PET) in AD [23]. This divergence can be explained by the stage of the affliction and the regional location in the brain. CSF sTREM2, a marker of microglial activation, is also increased in Advertizing patients compared with the healthy controls [25]. Still, several studies on sTREM2 have contradictory findings and the discriminative power of sTREM2 for Advertising is low. Further studies are required to establish the utility of sTREM2 in clinical practice [seventy].
Pro-inflammatory reactive astrocyte phenotypes take been linked to synaptic degeneration and glutamate dysregulation [13, 41]. Knockout of astrocytic glutamate transporters EAAT1 (glutamate/aspartate transporter, GLAST) and EAAT2 (glutamate transporter-1, GLT-ane) caused excitotoxicity and synaptic hyperexcitability in an AD model [71,72,73]. In the Advertizing mouse model, calcineurin (Catwo+/calmodulin-dependent phosphatase), a nuclear factor of the activated T-4 prison cell signaling pathway, has been constitute to link between astrocyte activation and hyperexcitability during Advertizing [72]. The astrocytes alter their function and morphology during AD, and may have different functions as AD progresses [43].
Most clinical trials for anti-inflammatory drugs in AD patients, including aspirin, prednisone, naproxen, diclofenac, indomethacin, and celecoxib, have failed to show definite improvements, even revealing some detrimental effects [74,75,76,77,78,79], although some have shown modest beneficial results. A subgroup assay showed that mild-to-moderate Advertisement patients who were APOE ε4 carriers benefitted from ibuprofen over the 12-month trial duration [eighty]. Another written report revealed protective effects of indomethacin in balmy-to-moderate Ad patients over 6 months, but the patient dropout charge per unit was loftier, which weakened the significance of the study [81]. The failure of the clinical trials may exist explained by the possible impact of the anti-inflammatory drugs on the protective effects of glial cells, the selection of participants, and the curt-term follow-up menses. For example, the Alzheimer's Illness Anti-inflammatory Prevention Trial (Accommodate) enrolled former cognitively unimpaired individuals and evaluated the furnishings of naproxen and celecoxib on cognitive function [79]. Although the participants were family unit members of AD patients, we did not know if they had AD, and cognitive impairment may not have started in them; hence, microglia and astrocytes may have been more neuroprotective than pro-inflammatory. The COX-inhibiting anti-inflammatory drugs may reduce microglial activation [82]. Furthermore, 3 years of follow-up may not be long enough for revealing a difference, equally cognitive decline may non start in participants within 3 years. Minocycline, which can cross the BBB and inhibit pro-inflammatory microglia [83], showed beneficial effects on retentiveness harm acquired past Aβ and reversed the increase in various inflammatory cytokines in an animal model of Advertising [84]. Recently, the Minocycline in Alzheimer's Disease Efficacy (MADE) trial compared two different doses of minocycline and placebo in mild AD patients over 2 years, but failed to demonstrate a beneficial effect [85]. The complexity of the relationship between microglial activation and neurodegeneration and the minimal treatment effects of minocycline were considered as reasons for the negative issue [86]. CHF 5074 (CSP-1103) is a modulator of microglia that polarizes microglia from the neurotoxic to the neuroprotective phenotype [38]. A CHF 5074 trial assessing mild cognitive impairment following Advertisement is currently in phase II (NCT01421056). Cromolyn, which is used in patients with asthma, has been found to induce neuroprotective microglial activation, promote Aβ42 uptake in microglia, and reduce the aggregation-prone Aβ levels [39]. The cromolyn is currently under a phase III trial (NCT02547818) every bit a therapeutic for early stages of AD.
PD
PD is the near frequent movement disorder and the 2nd most frequent neurodegenerative disease after AD [6]. The accumulation of Lewy bodies, intracellular inclusions that contain α-synuclein, and dopaminergic neuronal death in the substantia nigra pars compacta and other encephalon regions are neuropathological hallmarks of PD [6, 87]. In add-on, the activation of glial cells, including microglia and astrocytes, also contributes to the pathogenesis of PD. Several proteins that are encoded by genes associated with familial forms of PD, including α-synuclein (PARK1 and PARK4), parkin (PARK2), DJ-1 (PARK7), and ATPase 13A2 (ATP13A2 gene), are involved in the regulation of microglial and astrocyte activation [6, 41].
The incremental activation of microglial cells (MHC-Two-, ICAM-1-, and LFA01-positive cells) is observed in the substantia nigra of PD patients [88]. Furthermore, the degree of microglial activation is correlated with the dopaminergic concluding loss in early PD [89]. The activated microglia that environment dopaminergic neurons are generally pro-inflammatory [28]. The aggregated α-synuclein is released from dying dopaminergic neurons, and it activates microglia into the pro-inflammatory phenotype [90]. Over-expression of α-synuclein drives microglia into a reactive pro-inflammatory phenotype, and TNF-α, NO, and IL-1β derived from the pro-inflammatory microglia tin can modulate the neuroinflammatory process in PD [91, 92]. one-Methyl-4-phenyl-1,2,iii,6-tetrahydropyridine is known to cause dopaminergic neuron injury via mitochondrial dysfunction and by indirectly activating microglia [28, 93]. Lipopolysaccharide, a ligand of TLRs, can likewise cause dopaminergic neuronal expiry by activating the pro-inflammatory phenotype of microglia [28]. Jmjd3 has been reported to exist essential for the expression of the M2 microglial phenotype [35]. Suppression of Jmjd3 attenuates the neuroprotective polarization and over-activates the pro-inflammatory microglial response with the exacerbation of dopaminergic neuronal cell death in a PD mouse model [35]. However, the role of the neuroprotective microglial phenotype is still unclear.
Reactive astrocytes have been detected in the substantia nigra pars compacta of PD patients [94]. Astrocyte dysfunction plays a office in dopaminergic neurodegeneration. Various genes are involved in the development of PD and astrocyte biology [95], including PARK7 (encoding DJ-1), SNCA (encoding α-synuclein), PARK2 (encoding Parkin), PLA2G6 (encoding Ca2+-independent phospholipase Aii), ATP13A2 (encoding lysosomal blazon 5 ATPase, ATP13A2), LRRK2 (encoding leucine-rich repeat kinase 2, LRRK2), GBA (encoding β-glucocerebrosidase, GCase), and PINK1 (encoding PTEN-induced putative kinase 1, PINK1) genes [95]. The DJ-1 protein regulates astrocyte activation through the IFN-γ and TLR4 signaling [41, 96]. Maintaining the ATP13A2 level could prevent the activation of the NLPR3 inflammasome [41, 95].
Various anti-inflammatory treatments such equally dexamethasone, ibuprofen, amantadine, minocycline, pituitary adenylate cyclase-activating peptide, vasoactive intestinal peptide, IL-10, and TGF-β have shown preventive effects on dopaminergic prison cell expiry in animal models [97,98,99,100,101,102,103]. However, the effects of anti-inflammatory drugs in PD patients are contradictory. Ane meta-analysis concluded that the nonsteroidal anti-inflammatory drugs (NSAIDs) may non modify the adventure of PD, with but ibuprofen seeming to take a modest protective effect [104]. Another meta-assay ended that NSAIDs, except aspirin, may have a protective effect on the risk of PD [105]. Minocycline, which showed a neuroprotective effect in several in vivo and in vitro studies, was unsuccessful in altering the course of early PD over 12 and eighteen months in a randomized clinical trial [106,107,108]. NLY01, a glucagon-like peptide-ane receptor agonist, was protective against dopaminergic neuronal loss and aberrant behavioral office in a desultory PD mouse model [109]. This neuroprotective effect was attributed to the inhibition of the conversion of astrocytes to the neurotoxic phenotype, which was mediated by microglia [109].
ALS
ALS, also called Lou Gehrig'south disease, is an adult-onset progressive neurodegenerative disease in which motor neurons are selectively affected [110]. The etiology of almost ALS patients remains unidentified. Simply less than 10% of cases are due to mutations of specific genes, including superoxidase dismutase one (SOD1), C9orf72, TDP43, and FUS [18]. Neuroinflammation is a pathological mechanism common to ALS patients with and without genetic mutations, which is characterized by the infiltration of activated microglia and astrocytes. The activated microglia and astrocytes that produce pro-inflammatory cytokines are upregulated in post-mortem tissues of ALS patients [41, 111, 112]. A PET study has demonstrated increases in activated microglia [xiC-(R)PK11195 PET] and astrocytes (11C-DED PET) in living ALS patients [113, 114]. In addition, the CSF sTREM2 level is significantly college in sporadic ALS patients with varying affliction severity than controls [115]. In item, the CSF sTREM2 level is highest in the early-stage ALS, and in late stage, higher levels of CSF sTREM2 are associated with slower illness progression [115]. Prolonged loftier levels of CSF sTREM2 may exist indicative of a neuroprotective phenotype.
Toxicity acquired by mutant SOD1, the nigh common form of inherited ALS, is mediated past directly damage that is incurred within the motor neurons, microglia, and astrocytes [110]. The activated pro-inflammatory microglia and astrocytes produce toxic factors that crusade the initial damage and disease progression. The G930A-SOD1 transgenic mouse model has demonstrated the ability of microglia to switch from the neuroprotective to the pro-inflammatory phenotype from the onset of the pathology [28, 116]. The SOD1-mutant microglia isolated from mice with early-stage ALS express higher levels of M2 microglia phenotype markers and lower levels of pro-inflammatory microglia markers, compared with the SOD1-mutant microglia isolated from mice with end-stage ALS [117]. Altogether, as ALS progresses, the function of neuroprotective microglia may decrease and the proportion of pro-inflammatory phenotypes may increment. The C3-expressing pro-inflammatory astrocytes and astrocytic NLRP3 inflammasomes have been found to be upregulated in mail service-mortem ALS patients [41, 46, 118]. The activation of astrocytes in ALS decreases their protective effects and increases their detrimental effects [119]. Astrocytes with SOD1 mutations accept been reported to release soluble factors toxic to motor neurons [120]. IL-1α, TNF-α, and C1q released from microglia bulldoze astrocytes to the neurotoxic phenotype, while reducing reactive astrocytes by inhibiting these factors attenuates the disease progression in the G93A-SOD1 mouse model [121]. Still, little is known most the neuroprotective phenotype of astrocytes in the pathogenesis of ALS.
The ablation of NOX3 or NF-kB improved motor neuron survival in the G930A-SOD1 transgenic mouse model [28, 122]. In add-on, the administration of minocycline in G930A-SOD1 transgenic mice selectively adulterate the expression of markers for the pro-inflammatory microglia, inhibited the upregulation of NF-κB in the principal culture of microglia, and delayed the pathogenesis [28, 123]. Cromolyn, which induced the activation of neuroprotective microglia in the Ad mouse model, demonstrated a neuroprotective effect in the G93A-SOD1 transgenic mouse model by delaying the affliction onset and reducing the motor impairment [124].
Recently, masitinib, an oral tyrosine kinase inhibitor, has shown benign effects in ALS patients over 48 weeks [125]. Masitinib reduces the microgliosis and the emergence of aberrant glial cells in the G93A-SOD1 transgenic mouse model [126]. Regulatory T-lymphocytes (Tregs) can augment IL-4 expression, induce the M2-phenotype microglia, and filibuster the progression of the disease [127]. Upregulation of Tregs can be achieved using dimethyl fumarate (Tecfidera), and a phase 2 trial of Tecfidera is being conducted in patients with sporadic ALS [128]. In add-on, the infusion of tocilizumab in ALS patients reduced neuroinflammation [129], and it is now in a stage 2 clinical trial in ALS patients (NCT02469896), results of which are expected to exist announced soon.
Conclusion
Nosotros reviewed the roles of neuroinflammation in neurodegenerative diseases, focusing on microglia and astrocytes. In addition, clinical or experimental studies on treatments associated with neuroinflammation in neurodegenerative diseases were discussed. A balance between pro-inflammatory and neuroprotective glial cells may be critical in the progression of neurodegenerative diseases. Moreover, it has been reported that the activated microglia and reactive astrocytes influence each other. Due to the complexity of microglia and astrocyte phenotypes and the diverse types of drugs, the stages of neurodegenerative diseases (more than pro-inflammatory than neuroprotective) and the conditions of patients (confirmed pathology of disease and likely to progress within few years) may be crucial for demonstrating the benefits of anti-inflammatory treatments in clinical trials. The functions of microglia and astrocytes at specific stages of specific diseases in specific patients demand to be identified. The next step for trials is to determine a standard method for evaluating each phenotype of microglia and astrocytes to standardize further evaluation.
Availability of data and materials
Not applicable.
Abbreviations
- 11C-DED:
-
elevenC-deuterium-Fifty-deprenyl
- Aβ:
-
Amyloid-beta
- Act 1:
-
Activator i
- Advert:
-
Alzheimer's illness
- ALS:
-
Amyotrophic lateral sclerosis
- Arg1:
-
Arginase ane
- BBB:
-
Blood-encephalon barrier
- BDNF:
-
Brain-derived neurotrophic factor
- Chi3l3:
-
Chitinase-iii-Similar-3
- CCL:
-
C-C motif chemokine ligand
- CSF:
-
Cerebrospinal fluid
- CXCL:
-
C-X-C motif chemokine ligand
- DAMPs:
-
damage-associated molecular patterns
- EAE:
-
Experimental autoimmune encephalomyelitis
- ER:
-
Oestrogen receptor
- Fzd1:
-
Frizzled class receptor 1
- GFAP:
-
Glial fibrillary acidic poly peptide
- GM-CSF:
-
Granulocyte-macrophage colony-stimulating factor
- IFN:
-
Interferon
- IGF-ane:
-
Insulin-similar growth factor ane
- IL:
-
Interleukin
- JAK/STAT:
-
Janus kinase/signal transducers and activators of transcription
- Jmjd3:
-
Jumonji domain containing 3
- LacCer:
-
Lactosylceraide
- LPS:
-
Lipopolysaccharide
- NFκB:
-
Nuclear gene κB
- NO:
-
Nitric oxide
- PAMP:
-
Pathogen-associated molecular pattern
- PD:
-
Parkinson'south disease
- PET:
-
Positron emission tomography
- SCI:
-
Spinal cord injury
- SOD1:
-
Superoxidase dismutase i
- STAT3:
-
Indicate transducer and activator of transcription iii
- sTREM2:
-
Soluble triggering receptor expressed on myeloid cells 2
- TGF:
-
Transforming growth cistron
- TLR:
-
Toll-similar receptor
- TNF:
-
Tumor necrosis factor
- TrkB:
-
Tropomyosin receptor kinase B
- Tregs:
-
Regulatory T cells
References
-
Cova I, Markova A, Campini I, Grande Thousand, Mariani C, Pomati S. Worldwide trends in the prevalence of dementia. J Neurol Sci. 2017;379:259–sixty.
-
Jellinger KA. Basic mechanisms of neurodegeneration: a critical update. J Cell Mol Med. 2010;xiv:457–87.
-
Wyss-Coray T, Mucke 50. Inflammation in neurodegenerative illness--a double-edged sword. Neuron. 2002;35:419–32.
-
Kempuraj D, Thangavel R, Natteru PA, Selvakumar GP, Saeed D, Zahoor H, et al. Neuroinflammation induces neurodegeneration. J Neurol Neurosurg Spine. 2016;1:1003.
-
Russo MV, McGavern DB. Inflammatory neuroprotection following traumatic brain injury. Science. 2016;353:783–5.
-
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH. Mechanisms underlying inflammation in neurodegeneration. Cell. 2010;140:918–34.
-
Stephenson J, Nutma Eastward, van der Valk P, Amor Due south. Inflammation in CNS neurodegenerative diseases. Immunology. 2018;154:204–xix.
-
Subhramanyam CS, Wang C, Hu Q, Dheen ST. Microglia-mediated neuroinflammation in neurodegenerative diseases. Semin Jail cell Dev Biol. 2019;94:112–20.
-
Fields RD, Araque A, Johansen-Berg H, Lim SS, Lynch G, Nave KA, et al. Glial biology in learning and cognition. Neuroscientist. 2014;20:426–31.
-
Clarke LE, Barres BA. Emerging roles of astrocytes in neural circuit evolution. Nat Rev Neurosci. 2013;fourteen:311–21.
-
Luo XG, Chen SD. The changing phenotype of microglia from homeostasis to illness. Transl Neurodegener. 2012;1:nine.
-
Bachiller Southward, Jiménez-Ferrer I, Paulus A, Yang Y, Swanberg Grand, Deierborg T, et al. A microglia in neurological diseases: a road map to brain-affliction dependent-inflammatory response. Front end Prison cell Neurosci. 2018;12:488.
-
Liddelow SA, Barres BA. Reactive astrocytes: product, office, and therapeutic potential. Immunity. 2017;46:957–67.
-
De Biase LM, Schuebel KE, Fusfeld ZH, Jair K, Hawes IA, Cimbro R, et al. Local cues establish and maintain region-specific phenotypes of basal ganglia microglia. Neuron. 2017;95:341–56.
-
Heneka MT, Carson MJ, El Khoury J, Landreth GE, Brosseron F, Feinstein DL, et al. Neuroinflammation in Alzheimer's disease. Lancet Neurol. 2015;14:388–405.
-
Baufeld C, O'Loughlin E, Calcagno Due north, Madore C, Butovsky O. Differential contribution of microglia and monocytes in neurodegenerative diseases. J Neural Transm. 2018;125:809–26.
-
Lawson LJ, Perry VH, Dri P, Gordon S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience. 1990;39:151–70.
-
Hickman Southward, Izzy S, Sen P, Morsett Fifty, El Khoury J. Microglia in neurodegeneration. Nat Neurosci. 2018;21:1359–69.
-
Hickman SE, Kingery ND, Ohsumi TK, Borowsky ML, Wang LC, Ways TK, et al. The microglial sensome revealed by directly RNA sequencing. Nat Neurosci. 2013;xvi:1896–905.
-
Zhan Y, Paolicelli RC, Sforazzini F, Weinhard L, Bolasco M, Pagani F, et al. Deficient neuron-microglia signaling results in impaired functional brain connectivity and social behavior. Nat Neurosci. 2014;17:400–6.
-
Niraula A, Sheridan JF, Godbout JP. Microglia priming with aging and stress. Neuropsychopharmacology. 2017;42:318–33.
-
Scarf AM, Kassiou Yard. The translocator protein. J Nucl Med. 2011;52:677–eighty.
-
Malpetti Thousand, Kievit RA, Passamonti L, Jones PS, Tsvetanov KA, Rittman T, et al. Microglial activation and tau burden predict cognitive decline in Alzheimer'southward disease. Brain. 2020;143:1588–602.
-
Kwon HS, Lee EH, Park HH, Jin JH, Choi H, Lee KY, et al. Early on increment of soluble triggering receptor expressed on myeloid cells 2 in plasma might be a predictor of poor effect afterward ischemic stroke. J Clin Neurosci. 2020;73:215–8.
-
Bekris LM, Khrestian M, Dyne East, Shao Y, Pillai JA, Rao SM, et al. Soluble TREM2 and biomarkers of central and peripheral inflammation in neurodegenerative disease. J Neuroimmunol. 2018;319:19–27.
-
Suárez-Calvet Thousand, Kleinberger G, Araque Caballero M, Brendel Yard, Rominger A, Alcolea D, et al. sTREM2 cerebrospinal fluid levels are a potential biomarker for microglia activity in early-stage Alzheimer's affliction and associate with neuronal injury markers. EMBO Mol Med. 2016;eight:466–76.
-
Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012;122:787–95.
-
Tang Y, Le W. Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol Neurobiol. 2016;53:1181–94.
-
Zhao W, Xie W, Xiao Q, Beers DR, Appel SH. Protective effects of an anti-inflammatory cytokine, interleukin-four, on motoneuron toxicity induced past activated microglia. J Neurochem. 2006;99:1176–87.
-
Park KW, Lee DY, Joe EH, Kim SU, Jin BK. Neuroprotective office of microglia expressing interleukin-4. J Neurosci Res. 2005;81:397–402.
-
Ruckh JM, Zhao JW, Shadrach JL, van Wijngaarden P, Rao TN, Wagers AJ, et al. Rejuvenation of regeneration in the aging cardinal nervous organisation. Prison cell Stem Prison cell. 2012;10:96–103.
-
Lumeng CN, Bodzin JL, Saltiel AR. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest. 2007;117:175–84.
-
Odegaard JI, Ricardo-Gonzalez RR, Goforth MH, Morel CR, Subramanian V, Mukundan L, et al. Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature. 2007;447:1116–20.
-
Zhang H, Li Y, Yu J, Guo M, Meng J, Liu C, et al. Rho kinase inhibitor fasudil regulates microglia polarization and function. Neuroimmunomodulation. 2013;xx:313–22.
-
Tang Y, Li T, Li J, Yang J, Liu H, Zhang XJ, et al. Jmjd3 is essential for the epigenetic modulation of microglia phenotypes in the immune pathogenesis of Parkinson's disease. Prison cell Death Differ. 2014;21:369–80.
-
Kim SH, Noh MY, Kim HJ, Oh KW, Park J, Lee S, et al. A therapeutic strategy for Alzheimer's disease focused on allowed–inflammatory modulation. Dement Neurocogn Disord. 2019;18:33–46.
-
Miao H, Li R, Han C, Lu Ten, Zhang H. Minocycline promotes posthemorrhagic neurogenesis via M2 microglia polarization via upregulation of the TrkB/BDNF pathway in rats. J Neurophysiol. 2018;120:1307–17.
-
Porrini V, Lanzillotta A, Branca C, Benarese M, Parrella E, Lorenzini L, et al. CHF5074 (CSP-1103) induces microglia alternative activation in plaque-free Tg2576 mice and primary glial cultures exposed to beta-amyloid. Neuroscience. 2015;302:112–20.
-
Zhang C, Griciuc A, Hudry E, Wan Y, Quinti L, Ward J, et al. Cromolyn reduces levels of the Alzheimer'due south disease-associated amyloid beta-protein by promoting microglial phagocytosis. Sci Rep. 2018;8:1144.
-
Colombo E, Farina C. Astrocytes: primal regulators of neuroinflammation. Trends Immunol. 2016;37:608–twenty.
-
Oksanen M, Lehtonen S, Jaronen G, Goldsteins G, Hamalainen RH, Koistinaho J. Astrocyte alterations in neurodegenerative pathologies and their modeling in human induced pluripotent stem jail cell platforms. Cell Mol Life Sci. 2019;76:2739–threescore.
-
Sofroniew MV. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 2009;32:638–47.
-
Carter SF, Herholz One thousand, Rosa-Neto P, Pellerin L, Nordberg A, Zimmer ER. Astrocyte biomarkers in Alzheimer's disease. Trends Mol Med. 2019;25:77–95.
-
Mayo Fifty, Trauger SA, Blain G, Nadeau M, Patel B, Alvarez JI, et al. Regulation of astrocyte activation by glycolipids drives chronic CNS inflammation. Nat Med. 2014;20:1147–56.
-
Saijo K, Winner B, Carson CT, Collier JG, Boyer L, Rosenfeld MG, et al. A Nurr1/CoREST pathway in microglia and astrocytes protects dopaminergic neurons from inflammation-induced decease. Prison cell. 2009;137:47–59.
-
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, et al. Neurotoxic reactive astrocytes are induced past activated microglia. Nature. 2017;541:481–7.
-
Qian Y, Liu C, Hartupee J, Altuntas CZ, Gulen MF, Jane-Wit D, et al. The adaptor Act1 is required for interleukin 17-dependent signaling associated with autoimmune and inflammatory illness. Nat Immunol. 2007;8:247–56.
-
Colombo Due east, Cordiglieri C, Melli G, Newcombe J, Krumbholz 1000, Parada LF, et al. Stimulation of the neurotrophin receptor TrkB on astrocytes drives nitric oxide production and neurodegeneration. J Exp Med. 2012;209:521–35.
-
Haroon F, Drogemuller M, Handel U, Brunn A, Reinhold D, Nishanth 1000, et al. Gp130-dependent astrocytic survival is critical for the control of autoimmune central nervous system inflammation. J Immunol. 2011;186:6521–31.
-
Drogemuller K, Helmuth U, Brunn A, Sakowicz-Burkiewicz Chiliad, Gutmann DH, Mueller Due west, et al. Astrocyte gp130 expression is disquisitional for the control of toxoplasma encephalitis. J Immunol. 2008;181:2683–93.
-
Cekanaviciute Due east, Fathali N, Doyle KP, Williams AM, Han J, Buckwalter MS. Astrocytic transforming growth factor-beta signaling reduces subacute neuroinflammation after stroke in mice. Glia. 2014;62:1227–40.
-
Cekanaviciute E, Dietrich HK, Axtell RC, Williams AM, Egusquiza R, Wai KM, et al. Astrocytic TGF-beta signaling limits inflammation and reduces neuronal damage during cardinal nervous system toxoplasma infection. J Immunol. 2014;193:139–49.
-
Hindinger C, Bergmann CC, Hinton DR, Phares TW, Parra GI, Hussain Due south, et al. IFN-gamma signaling to astrocytes protects from autoimmune mediated neurological inability. PLoS One. 2012;7:e42088.
-
Tiwari-Woodruff S, Morales LB, Lee R, Voskuhl RR. Differential neuroprotective and antiinflammatory effects of estrogen receptor (ER) blastoff and ERbeta ligand treatment. Proc Natl Acad Sci U South A. 2007;104:14813–viii.
-
Okada S, Nakamura G, Katoh H, Miyao T, Shimazaki T, Ishii K, et al. Conditional ablation of Stat3 or Socs3 discloses a dual office for reactive astrocytes afterwards spinal cord injury. Nat Med. 2006;12:829–34.
-
Islam O, Loo TX, Heese K. Brain-derived neurotrophic factor (BDNF) has proliferative effects on neural stalk cells through the truncated TRK-B receptor, MAP kinase, AKT, and STAT-3 signaling pathways. Curr Neurovasc Res. 2009;vi:42–53.
-
Herrmann JE, Imura T, Song B, Qi J, Ao Y, Nguyen TK, et al. STAT3 is a critical regulator of astrogliosis and scar germination after spinal cord injury. J Neurosci. 2008;28:7231–43.
-
Ceyzeriat 1000, Abjean Fifty, Carrillo-de Sauvage MA, Ben Haim L, Escartin C. The complex STATes of astrocyte reactivity: how are they controlled by the JAK-STAT3 pathway? Neuroscience. 2016;330:205–18.
-
Prince M, Ali GC, Guerchet M, Prina AM, Albanese Due east, Wu YT. Recent global trends in the prevalence and incidence of dementia, and survival with dementia. Alzheimers Res Ther. 2016;eight:23.
-
Tarkowski E, Andreasen Northward, Tarkowski A, Blennow K. Intrathecal inflammation precedes development of Alzheimer'due south disease. J Neurol Neurosurg Psychiatry. 2003;74:1200–5.
-
Brosseron F, Krauthausen M, Kummer M, Heneka MT. Body fluid cytokine levels in mild cerebral impairment and Alzheimer's disease: a comparative overview. Mol Neurobiol. 2014;l:534–44.
-
Yan LJ, Xiao Yard, Chen R, Cai Z. Metabolic dysfunction of astrocyte: an initiating factor in beta-amyloid pathology? Crumbling Neurodegener. 2013;1:7–fourteen.
-
Takata K, Kitamura Y, Saeki K, Terada Thou, Kagitani S, Kitamura R, et al. Galantamine-induced amyloid-{beta} clearance mediated via stimulation of microglial nicotinic acetylcholine receptors. J Biol Chem. 2010;285:40180–91.
-
Asai H, Ikezu S, Tsunoda S, Medalla M, Luebke J, Haydar T, et al. Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nat Neurosci. 2015;18:1584–93.
-
Mawuenyega KG, Sigurdson W, Ovod Five, Munsell 50, Kasten T, Morris JC, et al. Decreased clearance of CNS beta-amyloid in Alzheimer's disease. Science. 2010;330:1774.
-
Jimenez Due south, Baglietto-Vargas D, Caballero C, Moreno-Gonzalez I, Torres M, Sanchez-Varo R, et al. Inflammatory response in the hippocampus of PS1M146L/APP751SL mouse model of Alzheimer's disease: age-dependent switch in the microglial phenotype from alternative to archetype. J Neurosci. 2008;28:11650–61.
-
Lee DC, Rizer J, Selenica ML, Reid P, Kraft C, Johnson A, et al. LPS- induced inflammation exacerbates phospho-tau pathology in rTg4510 mice. J Neuroinflammation. 2010;7:56.
-
Fan Z, Brooks DJ, Okello A, Edison P. An early and late pinnacle in microglial activation in Alzheimer's disease trajectory. Encephalon. 2017;140:792–803.
-
Varnum MM, Ikezu T. The classification of microglial activation phenotypes on neurodegeneration and regeneration in Alzheimer's disease brain. Arch Immunol Ther Exp (Warsz). 2012;60:251–66.
-
Carmona S, Zahs M, Wu E, Dakin K, Bras J, Guerreiro R. The role of TREM2 in Alzheimer'south disease and other neurodegenerative disorders. Lancet Neurol. 2018;17:721–30.
-
Rothstein JD, Dykes-Hoberg M, Pardo CA, Bristol LA, Jin L, Kuncl RW, et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron. 1996;16:675–86.
-
Sompol P, Furman JL, Pleiss MM, Kraner SD, Artiushin IA, Batten SR, et al. Calcineurin/NFAT signaling in activated astrocytes drives network hyperexcitability in Abeta-bearing mice. J Neurosci. 2017;37:6132–48.
-
Kellner V, Menkes-Caspi N, Beker S, Stern EA. Amyloid-beta alters ongoing neuronal activity and excitability in the frontal cortex. Neurobiol Aging. 2014;35:1982–91.
-
Aisen PS, Davis KL, Berg JD, Schafer Thou, Campbell Chiliad, Thomas RG, et al. A randomized controlled trial of prednisone in Alzheimer'southward illness. Alzheimer's disease cooperative written report. Neurology. 2000;54:588–93.
-
Aisen PS, Schafer KA, Grundman M, Pfeiffer E, Sano Thou, Davis KL, et al. Furnishings of rofecoxib or naproxen vs placebo on Alzheimer disease progression: a randomized controlled trial. JAMA. 2003;289:2819–26.
-
Bentham P, Gray R, Sellwood E, Hills R, Crome P, Raftery J. Aspirin in Alzheimer's affliction (AD2000): a randomised open-label trial. Lancet Neurol. 2008;7:41–9.
-
Scharf Southward, Mander A, Ugoni A, Vajda F, Christophidis North. A double-bullheaded, placebo-controlled trial of diclofenac/misoprostol in Alzheimer's illness. Neurology. 1999;53:197–201.
-
Thal LJ, Ferris SH, Kirby L, Block GA, Lines CR, Yuen Due east, et al. A randomized, double-blind, study of rofecoxib in patients with mild cognitive damage. Neuropsychopharmacology. 2005;30:1204–fifteen.
-
Martin BK, Szekely C, Brandt J, Piantadosi S, Breitner JC, Craft S, et al. Cognitive office over time in the Alzheimer'south disease anti-inflammatory prevention trial (Accommodate): results of a randomized, controlled trial of naproxen and celecoxib. Curvation Neurol. 2008;65:896–905.
-
Pasqualetti P, Bonomini C, Dal Forno G, Paulon L, Sinforiani East, Marra C, et al. A randomized controlled study on effects of ibuprofen on cerebral progression of Alzheimer'due south disease. Aging Clin Exp Res. 2009;21:102–10.
-
Rogers J, Kirby LC, Hempelman SR, Berry DL, McGeer PL, Kaszniak AW, et al. Clinical trial of indomethacin in Alzheimer's disease. Neurology. 1993;43:1609–eleven.
-
Krause DL, Müller N. Neuroinflammation, microglia and implications for anti-inflammatory handling in Alzheimer's illness. Int J Alzheimers Dis. 2010;2010:732806.
-
Familian A, Boshuizen RS, Eikelenboom P, Veerhuis R. Inhibitory upshot of minocycline on amyloid beta fibril formation and man microglial activation. Glia. 2006;53:233–xl.
-
Garcez ML, Mina F, Bellettini-Santos T, Carneiro FG, Luz AP, Schiavo GL, et al. Minocycline reduces inflammatory parameters in the brain structures and serum and reverses retention impairment caused by the administration of amyloid beta (ane-42) in mice. Prog Neuropsychopharmacol Biol Psychiatry. 2017;77:23–31.
-
Howard R, Zubko O, Bradley R, Harper East, Pank L, O'Brien J, et al. Minocycline at two different dosages vs placebo for patients with mild Alzheimer disease: a randomized clinical trial. JAMA Neurol. 2019;77:164–74.
-
Gyengesi East, Munch G. In search of an anti-inflammatory drug for Alzheimer disease. Nat Rev Neurol. 2020;16:131–ii.
-
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak Due east. Staging of encephalon pathology related to sporadic Parkinson's disease. Neurobiol Aging. 2003;24:197–211.
-
Rocha NP, de Miranda Every bit, Teixeira AL. Insights into neuroinflammation in Parkinson's disease: from biomarkers to anti-inflammatory based therapies. Biomed Res Int. 2015;2015:628192.
-
Ouchi Y, Yoshikawa E, Sekine Y, Futatsubashi M, Kanno T, Ogusu T, et al. Microglial activation and dopamine terminal loss in early Parkinson'southward disease. Ann Neurol. 2005;57:168–75.
-
Zhang Westward, Wang T, Pei Z, Miller DS, Wu X, Block ML, et al. Aggregated blastoff-synuclein activates microglia: a process leading to disease progression in Parkinson's affliction. FASEB J. 2005;19:533–42.
-
Rojanathammanee L, Irish potato EJ, Combs CK. Expression of mutant alpha-synuclein modulates microglial phenotype in vitro. J Neuroinflammation. 2011;8:44.
-
Hirsch EC, Hunot Southward. Neuroinflammation in Parkinson'south disease: a target for neuroprotection? Lancet Neurol. 2009;8:382–97.
-
Dauer W, Przedborski S. Parkinson'south affliction: mechanisms and models. Neuron. 2003;39:889–909.
-
Miklossy J, Doudet DD, Schwab C, Yu S, McGeer EG, McGeer PL. Role of ICAM-1 in persisting inflammation in Parkinson disease and MPTP monkeys. Exp Neurol. 2006;197:275–83.
-
Booth HDE, Hirst WD, Wade-Martins R. The role of astrocyte dysfunction in Parkinson's disease pathogenesis. Trends Neurosci. 2017;40:358–lxx.
-
Kim JH, Choi DJ, Jeong HK, Kim J, Kim DW, Choi SY, et al. DJ-1 facilitates the interaction between STAT1 and its phosphatase, SHP-1, in brain microglia and astrocytes: a novel anti-inflammatory function of DJ-1. Neurobiol Dis. 2013;60:one–10.
-
Manthripragada AD, Schernhammer ES, Qiu J, Friis S, Wermuth L, Olsen JH, et al. Not-steroidal anti-inflammatory drug use and the risk of Parkinson'due south affliction. Neuroepidemiology. 2011;36:155–61.
-
Gao X, Chen H, Schwarzschild MA, Ascherio A. Use of ibuprofen and risk of Parkinson disease. Neurology. 2011;76:863–9.
-
Malagelada C, Jin ZH, Jackson-Lewis V, Przedborski Due south, Greene LA. Rapamycin protects against neuron death in in vitro and in vivo models of Parkinson's illness. J Neurosci. 2010;xxx:1166–75.
-
Delgado M, Jonakait GM, Ganea D. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide inhibit chemokine product in activated microglia. Glia. 2002;39:148–61.
-
Lamine A, Letourneau Thou, Doan ND, Maucotel J, Couvineau A, Vaudry H, et al. Characterizations of a synthetic pituitary adenylate cyclase-activating polypeptide analog displaying potent neuroprotective activity and reduced in vivo cardiovascular side furnishings in a Parkinson's affliction model. Neuropharmacology. 2016;108:440–50.
-
Qian L, Block ML, Wei SJ, Lin CF, Reece J, Pang H, et al. Interleukin-10 protects lipopolysaccharide-induced neurotoxicity in principal midbrain cultures by inhibiting the function of NADPH oxidase. J Pharmacol Exp Ther. 2006;319:44–52.
-
Zhu Y, Yang GY, Ahlemeyer B, Pang L, Che XM, Culmsee C, et al. Transforming growth factor-beta 1 increases bad phosphorylation and protects neurons against damage. J Neurosci. 2002;22:3898–909.
-
Samii A, Etminan M, Wiens MO, Jafari S. NSAID use and the take a chance of Parkinson's affliction: systematic review and meta-analysis of observational studies. Drugs Aging. 2009;26:769–79.
-
Gagne JJ, Ability MC. Anti-inflammatory drugs and risk of Parkinson disease: a meta-analysis. Neurology. 2010;74:995–1002.
-
NINDS Internet-PD Investigators. A randomized, double-blind, futility clinical trial of creatine and minocycline in early Parkinson disease. Neurology. 2006;66:664–71.
-
NINDS Internet-PD Investigators. A pilot clinical trial of creatine and minocycline in early on Parkinson disease: 18-calendar month results. Clin Neuropharmacol. 2008;31:141–50.
-
Cankaya Southward, Cankaya B, Kilic U, Kilic Due east, Yulug B. The therapeutic function of minocycline in Parkinson's disease. Drugs Context. 2019;8:212553.
-
Yun SP, Kam TI, Panicker Northward, Kim S, Oh Y, Park JS, et al. Block of A1 astrocyte conversion by microglia is neuroprotective in models of Parkinson's affliction. Nat Med. 2018;24:931–8.
-
Boillee Due south, Vande Velde C, Cleveland DW. ALS: a disease of motor neurons and their nonneuronal neighbors. Neuron. 2006;52:39–59.
-
Henkel JS, Engelhardt JI, Siklos Fifty, Simpson EP, Kim SH, Pan T, et al. Presence of dendritic cells, MCP-1, and activated microglia/macrophages in amyotrophic lateral sclerosis spinal cord tissue. Ann Neurol. 2004;55:221–35.
-
Kawamata T, Akiyama H, Yamada T, McGeer PL. Immunologic reactions in amyotrophic lateral sclerosis brain and spinal cord tissue. Am J Pathol. 1992;140:691–707.
-
Turner MR, Cagnin A, Turkheimer FE, Miller CC, Shaw CE, Brooks DJ, et al. Evidence of widespread cognitive microglial activation in amyotrophic lateral sclerosis: an [11C](R)-PK11195 positron emission tomography study. Neurobiol Dis. 2004;fifteen:601–9.
-
Johansson A, Engler H, Blomquist G, Scott B, Wall A, Aquilonius SM, et al. Evidence for astrocytosis in ALS demonstrated by [11C](L)-deprenyl-D2 PET. J Neurol Sci. 2007;255:17–22.
-
Cooper-Knock J, Greenish C, Altschuler One thousand, Wei W, Bury JJ, Heath PR, et al. A data-driven approach links microglia to pathology and prognosis in amyotrophic lateral sclerosis. Acta Neuropathol Commun. 2017;five:23.
-
Hensley K, Fedynyshyn J, Ferrell South, Floyd RA, Gordon B, Grammas P, et al. Message and protein-level acme of tumor necrosis factor blastoff (TNF alpha) and TNF blastoff-modulating cytokines in spinal cords of the G93A-SOD1 mouse model for amyotrophic lateral sclerosis. Neurobiol Dis. 2003;14:74–80.
-
Liao B, Zhao Westward, Beers DR, Henkel JS, Appel SH. Transformation from a neuroprotective to a neurotoxic microglial phenotype in a mouse model of ALS. Exp Neurol. 2012;237:147–52.
-
Johann S, Heitzer M, Kanagaratnam M, Goswami A, Rizo T, Weis J, et al. NLRP3 inflammasome is expressed by astrocytes in the SOD1 mouse model of ALS and in human sporadic ALS patients. Glia. 2015;63:2260–73.
-
Yamanaka M, Komine O. The multi-dimensional roles of astrocytes in ALS. Neurosci Res. 2018;126:31–8.
-
Nagai M, Re DB, Nagata T, Chalazonitis A, Jessell TM, Wichterle H, et al. Astrocytes expressing ALS-linked mutated SOD1 release factors selectively toxic to motor neurons. Nat Neurosci. 2007;x:615–22.
-
Guttenplan KA, Weigel MK, Adler DI, Couthouis J, Liddelow SA, Gitler AD, et al. Knockout of reactive astrocyte activating factors slows disease progression in an ALS mouse model. Nat Commun. 2020;eleven:3753.
-
Frakes AE, Ferraiuolo L, Haidet-Phillips AM, Schmelzer L, Braun Fifty, Miranda CJ, et al. Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron. 2014;81:1009–23.
-
Kobayashi K, Imagama S, Ohgomori T, Hirano G, Uchimura Grand, Sakamoto K, et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Expiry Dis. 2013;4:e525.
-
Granucci EJ, Griciuc A, Mueller KA, Mills AN, Le H, Dios AM, et al. Cromolyn sodium delays affliction onset and is neuroprotective in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Sci Rep. 2019;9:17728.
-
Mora JS, Genge A, Chio A, Estol CJ, Chaverri D, Hernandez Thousand, et al. Masitinib as an improver therapy to riluzole in patients with amyotrophic lateral sclerosis: a randomized clinical trial. Amyotroph Lateral Scler Frontotemporal Degener. 2020;21:five–14.
-
Trias E, Ibarburu Due south, Barreto-Nunez R, Babdor J, Maciel TT, Guillo Thousand, et al. Mail-paralysis tyrosine kinase inhibition with masitinib abrogates neuroinflammation and slows disease progression in inherited amyotrophic lateral sclerosis. J Neuroinflammation. 2016;xiii:177.
-
Beers DR, Henkel JS, Zhao W, Wang J, Huang A, Wen Due south, et al. Endogenous regulatory T lymphocytes improve amyotrophic lateral sclerosis in mice and correlate with disease progression in patients with amyotrophic lateral sclerosis. Brain. 2011;134:1293–314.
-
Vucic Due south, Ryder J, Mekhael L, Rd H, Mathers S, Needham M, et al. Phase 2 randomized placebo controlled double blind study to assess the efficacy and safety of tecfidera in patients with amyotrophic lateral sclerosis (TEALS study): study protocol clinical trial (SPIRIT compliant). Medicine. 2020;99:e18904.
-
Mizwicki MT, Fiala M, Magpantay 50, Aziz North, Sayre J, Liu G, et al. Tocilizumab attenuates inflammation in ALS patients through inhibition of IL6 receptor signaling. Am J Neurodegener Dis. 2012;1:305–xv.
Acknowledgments
Nosotros would like to thank Editage (world wide web.editage.co.kr) for English linguistic communication editing.
Funding
This review was supported by the Basic Scientific discipline Research Program of the National Enquiry Foundation of Korea, which was funded by the Ministry of Scientific discipline, ICT, and Future Planning (2018R1A2A2A15023219), a grant of the Korea Health Technology R&D Project through the Korea Health Industry Evolution Plant (KHIDI) funded by the Ministry of Health & Welfare, Republic of korea (HI20C0253), and the Medical Inquiry Middle (2017R1A5A2015395).
Author information
Affiliations
Contributions
KSH conceived the idea of this review. KHS drafted the manuscript and created the figures. KHS and KSH performed the literature search and reviewed the content of this manuscript. The authors read and approved the final manuscript.
Author's information
Not applicable.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicative.
Consent for publication
Non applicable.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits utilize, sharing, adaptation, distribution and reproduction in any medium or format, equally long every bit y'all give appropriate credit to the original author(s) and the source, provide a link to the Artistic Eatables licence, and betoken if changes were fabricated. The images or other third party material in this commodity are included in the article'southward Creative Eatables licence, unless indicated otherwise in a credit line to the material. If textile is not included in the article's Creative Eatables licence and your intended employ is non permitted by statutory regulation or exceeds the permitted use, you volition demand to obtain permission straight from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Artistic Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made bachelor in this article, unless otherwise stated in a credit line to the data.
Reprints and Permissions
About this article
Cite this article
Kwon, H.S., Koh, SH. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener 9, 42 (2020). https://doi.org/10.1186/s40035-020-00221-ii
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/ten.1186/s40035-020-00221-2
Keywords
- Neuroinflammation
- Neurodegenerative diseases
- Microglia
- Astrocytes
Source: https://translationalneurodegeneration.biomedcentral.com/articles/10.1186/s40035-020-00221-2
0 Response to "Letã¢â‚¬â„¢s Make Microglia Great Again in Neurodegenerative Disorders"
Post a Comment